제품특징
Advantages
- 장 오가노이드를 단층(monolayer) 및 ALI (Air-Liquid Interface) 배양으로 전환하여 apical surface에 쉽게 접근 가능
- 실험 전반에 걸쳐 일관된 결과 생성
IntestiCult Organoid Growth Medium (OGM)와 Organoid Differentiation Medium (ODM) 비교
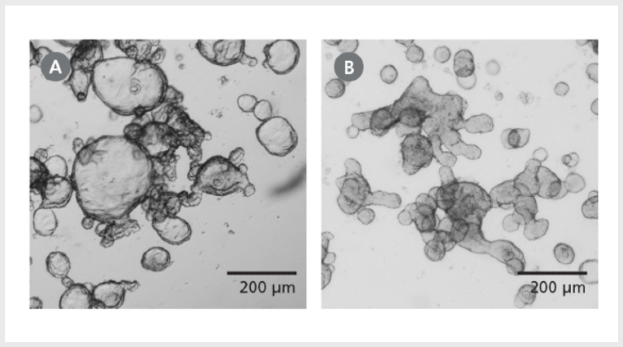
Figure 1. Differentiated Human Intestinal Organoids Display a Budded Morphology
(A) Organoids grown in IntestiCult™ OGM are primarily cystic. (B) When switched to IntestiCult™ ODM, organoids develop a thickened epithelium with a pronounced, budded morphology indicative of a more differentiated state. Organoids were imaged on day 5 of expansion or differentiation respectively.
Organoid 형성 후 분석: epithelial layer markers (MUC2, CHGA, KRAT20)발현 확인
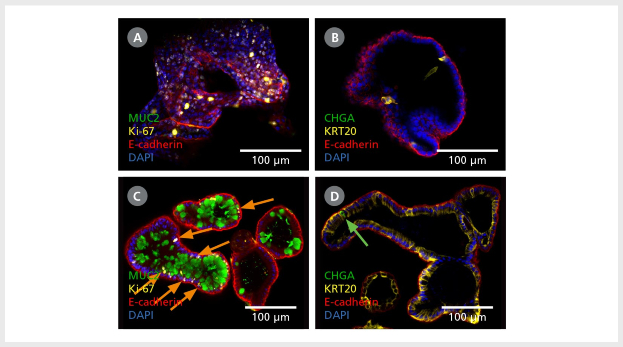
Figure 2. Intestinal Organoids Contain a Higher Proportion of Mature Cell Types Following Differentiation in IntestiCult™ ODM
(A, B) Organoids grown in IntestiCult™ OGM are enriched for Ki-67+ proliferative cells (A), while containing few differentiated cell types such as goblet cells (MUC2, A), enterocytes (KRT20, B), and enteroendocrine cells (CHGA, B). (C, D) When switched to IntestiCult™ ODM, organoids contain a small number of Ki-67+ proliferative cells (C, arrows), with more physiological proportions of goblet cells (MUC2, C), enterocytes (KRT20, D), and chromogranin A- (CHGA-)positive enteroendocrine cells (D, arrow).
Intestinal organoid의 단층 및 ALI (Air-Liquid Interface) 배양
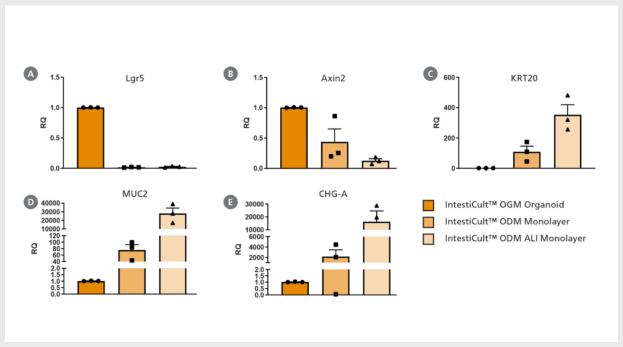
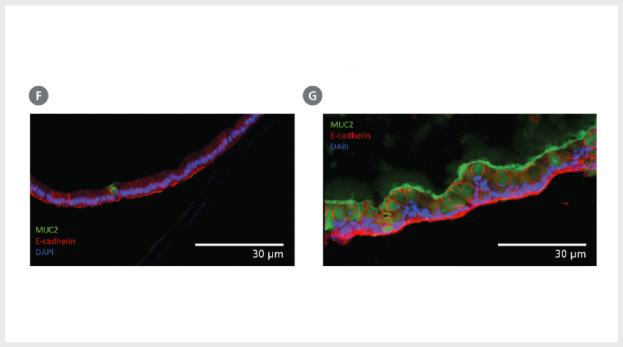
Figure 3. Differentiation of Intestinal Epithelium at the Air-Liquid Interface (ALI) Using IntestiCult™ ODM
(A – E) Growing organoid-derived monolayers as ALI cultures drives further differentiation of intestinal epithelial cultures as seen by changes in gene expression measured by RT-qPCR. Relative quantification (RQ) for each marker is shown relative to actB and TBP housekeeping genes and normalized with respect to undifferentiated organoids grown in IntestiCult OGM (Human). Progenitor markers (A) Lgr5 and (B) Axin2 are significantly reduced in both submerged monolayers and ALI cultures, while markers of enterocytes (KRT20, C), goblet cells (MUC2, D), and enteroendocrine cells (CHGA, E) are significantly increased. Further reduction in Axin2 is seen in ALI monolayers with an increase in expression of KRT20, MUC2, and CHGA. (F, G) Comparing cross-sections of organoid monolayers grown in IntestiCult™ ODM as (F) submerged culture or (G) at the ALI shows further differentiation of the intestinal epithelium with an increased proportion of goblet cells and extracellular mucus (MUC2, green).
단층(monolayer) 및 ALI (Air-Liquid Interface) 배양된 Intestinal organoid와 Caco-2 cell의 epithelial barrier 비교
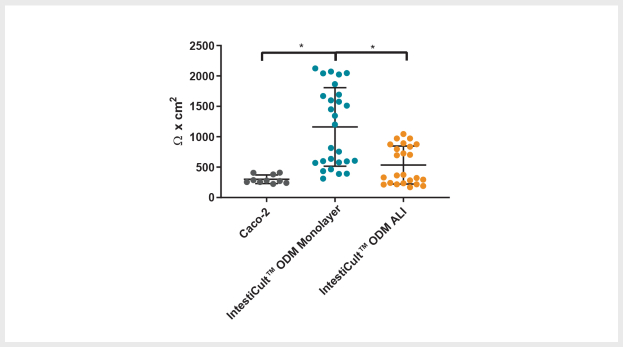
Figure 4. Differentiated Organoid-Derived Monolayers and ALI Cultures Display More Physiological Trans-Epithelial Electrical Resistance (TEER) than Caco-2 Cells
Differentiated organoid-derived monolayers grown as a submerged monolayer (IntestiCult™ ODM Monolayer), or at the ALI (IntestiCult™ ODM ALI), show higher TEER values as compared to Caco-2 cultures.Organoid-derived monolayers grown at the ALI show a loosening of tight junctions due to further differentiation of the brush border, and thus lower TEER values are observed. * p < 0.0001.
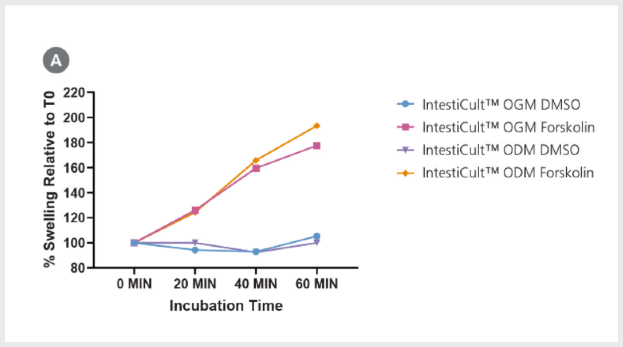
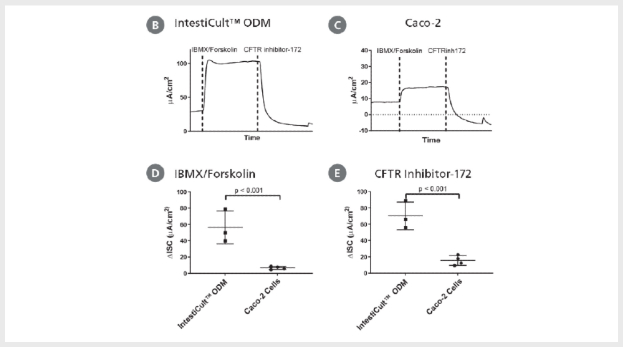
Figure 5. Differentiated Intestinal Organoids Provide a Suitable Model for Studying CFTR Response In Vitro
(A) Organoids differentiated further in IntestiCult™ ODM show a comparable degree of swelling when treated with forskolin as compared to organoids grown in IntestiCult™ OGM, demonstrating suitability for use in forskolin-induced swelling assays. (B – E) Ussing chamber analysis of submerged (B) organoid-derived monolayers and (C) Caco-2 cultures demonstrate increased sensitivity of organoid-derived monolayers to CFTR activation and inhibition by IBMX/Forskolin and CFTR Inhibitor-172 respectively. (D, E) Analysis of CFTR modulation by IBMX/Forskolin and CFTR Inhibitor-172 show significantly greater (D) activation and (E) inhibition of CFTR activity in organoid-derived monolayers as compared to Caco-2 cultures (p < 0.001 for both).
















 카트담기
카트담기 견적문의
견적문의